About the project
The aim of this international interdisciplinary project is to provide knowledge about the populations’ risk factors and health consequences of COVID-19. An additional aim is to provide information about how treatment with medications may modify the course and severity of COVID-19 disease. We will specifically study vulnerable patient groups (e.g. children, pregnant women and patients with co-morbidities like cancer) and assess mental health outcomes. To do so we will link several health care registries covering over 30 million individuals in Norway, Italy, Denmark and UK. We will incorporate our results in an online calculator that can be used to predict risk for severe COVID-19 disease.
The project will start 1. July 2020 and end 31. December 2025.
Objectives
It is imperative to fill our urgent knowledge gaps on epidemiological risk factors, course and consequences of COVID-19 in the general and potentially vulnerable population, including children, and to determine health outcomes of patients with the infection.
Understanding whether available pharmacotherapy options may modify the course and severity of COVID-19 using real-world pharmacoepidemiological data, is an impelling clinical question. The proposed project aims to fill part of these gaps, and will generate timely, methodologically sound evidence stemming from high-quality, detailed, and already available population-based data covering a population of over 30 million inhabitants in Norway, Italy, Denmark and Scotland. The specific objectives are divided in three work packages (WP), as illustrated in Figure 1 below.
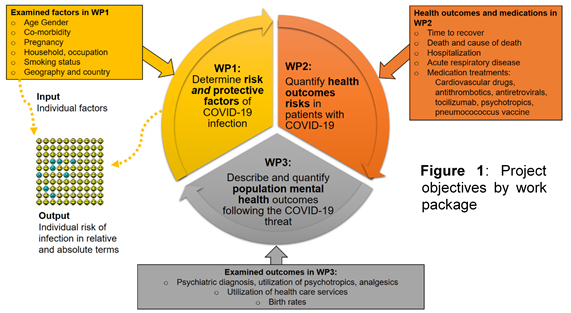
As part of the research output, we aim to develop an innovative electronic, risk algorithm that can be used to predict individual-specific risk for severe COVID-19 and prognosis, which is understandable to lay persons, and applicable in clinical setting, also in pediatrics.
Cooperation
EU-COVID-19 is a multinational project led by the Dpt. of Pharmacy, Faculty of Mathematics and Natural Sciences, University of Oslo (UiO). The project manager, Prof. Hedvig Nordeng, is head of the Pharmacoepidemiology and Drug Safety Research group at UiO.
Project group: Partners have specifically been selected based on their scientific expertise and experience. It includes experts in epidemiology, pharmacology, biostatistics, paediatrics, infectious diseases, signal detection and regulatory pharmacovigilance. All project members have extensive experience in conducting multi-database studies using advanced methods in biostatistics and epidemiology.
Vi skal bidra med forskning på COVID-19!
