PharmaTox PhD student Mari Roberts Spildrejorde will defend her thesis Dec 7, 2023.
News
Professor Hedvig Nordeng was awarded the Norwegian Women's Health Public Health Association's Women's Health Research Prize.
We proudly announce The PharmaTox Yearly Report 2022!
Please join us in congratulating Prof. Nordeng on her publication record for 2021.
Are you interested in how genetic and epigenetic data can be used to gain new insights in epidemiology?
We had a great group of UiO:Life Science students working with us this summer! They had backgrounds from biosciences, health economy, psychology, neuroscience, public health and informatics.
In June 2022 Johanne Naper Trønnes and Gerd Marie Harris defended their PhDs, and this summer their last papers were published!
PharmaTox-affiliated postdoc Nhung Trinh and PhD student Emilie Willoch Olstad attended the one-week summer course “Genetic and Epigenetic Epidemiology” in Florence, Italy, organised by the European Educational Programme in Epidemiology (EEPE). The course covered both fundamental knowledge and emerging methodologies within the field, presented by lecturers at the forefront of genetic and epigenetic epidemiological research.
Eimir Hurley presented the work that has been done to transform the Norwegian data to the OMOP Common Data Model.
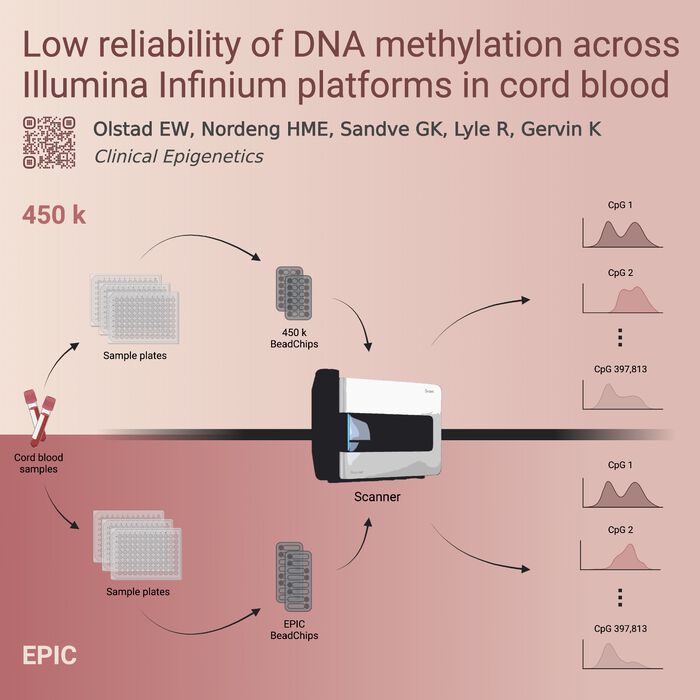
PharmaTox researchers have published a new article, "Low reliability of DNA methylation across Illumina Infinium platforms in cord blood: implications for replication studies and meta-analyses of prenatal exposures", in Clinical Epigenetics. Read it here.
PharmaTox researchers have published a new paper in Toxicology and Applied Pharmacology: "Paracetamol perturbs neuronal arborization and disrupts the cytoskeletal proteins SPTBN1 and TUBB3 in both human and chicken in vitro models". Read it here.
PharmaTox doctoral candidate Gerd Marie Eskerud Harris defends her thesis "Migraine pharmacotherapy during pregnancy: Utilization patterns and associations with child neurodevelopment" for the degree of Philosophiae Doctor.
PharmaTox researchers set out to answer the question "Is prenatal exposure to triptans associated with an increased risk of attention-deficit/hyperactivity disorder (ADHD) in offspring?".
Harald Eia lurer på om det er sånn at man kjører masse data inn i datamaksinen, og så finner man at barnet får tannstein dersom mor brukte briller i svangerskapet. Geir Kjetil Sandve svarer.
Da Kristina Gervin starter på det hun kaller en litt barnslig forklaring av epigenetikk, ber Harald Eia om den ikke-barnslige forklaringen. Han angrer seg umiddelbart.
The PharmaTox associated professor Eivind Ystrøm has been awarded the ERC Consolidator grant. Congratulations!
– Hvordan går du fram for å finne om det har en effekt på barna at mora tok mye smertestillende i svangerskapet? Om det er medisinbruk eller sårbare gener?, spør Harald Eia, og Eivind Ystrøm svarer.
The Department of Epidemiology, Biostatistics, and Occupational Health at McGill University/ Professor Robert Platt is one of our international partners in the iAPOGEE INTPART project. Vivian has received an iAPOGEE Scholarship to come and work with us on the treatment of cardiovascular diseases in pregnancy. Below she presents herself.
Hvordan påvirker mors medisinbruk hjernen til barnet i magen?, spør Harald Eia, og Hedvig Nordeng svarer.
PharmaTox PhD student Nils-Anders Labba has successfully defended his doctoral thesis «In vitro models and methods for assessment of developmental neurotoxicity and neuroprotection». Congratulations!
The PharmaTox yearly report of 2021 is now available! Download it here.
In the November issue of Nature Machine Intelligence, you can read about “immuneML”; an open-source software ecosystem developed to overcome the current limitations when using machine learning to identify patterns in adaptive immune receptor repertoires (AIRRs). “ImmuneML” was realised by great collaborative and multidisciplinary work, including several PharmaTox-affiliated researchers.
We are excited to announce that UiO:RealArt, led by PharmaTox professor Hedvig Nordeng in collaboration with a multidisciplinary team of researchers, has been appointed as a new convergence environment at the University of Oslo!
PharmaTox researchers represented at the NOFE meeting in Bergen, November 3 and 4, 2021.